
News & Events
News & Events
News
September 09, 2015
Hitachi and CiRA agree to collaborate in creating a "Healthy Volunteers' iPS Cell Panel"
Tokyo, Japan, September 7, 2015 — Hitachi, Ltd. (TSE:6501, "Hitachi") and the Center for iPS Cell* Research and Application (Director: Shinya Yamanaka; "CiRA") at Kyoto University have announced today that they have reached an agreement to collaborate in building a "healthy volunteers' iPS cell panel."
CiRA has been generating iPS Cells from the cells of patients suffering from a variety of diseases as part of research projects including "The Program for Intractable Diseases Research utilizing Disease-specific iPS cells" of the Program in the Research Center Network for Realization of Regenerative Medicine, funded by the Japan Agency for Medical Research and Development (AMED). By depositing the patient-specific iPS cells into public cell banks, CiRA has put in place an environment in which many researchers and companies are able to use the cells to advance research aimed at the elucidating the onset mechanisms for intractable diseases and the development of new drugs.
The research requires comparative studies of iPS cells from persons who have the disease in question and those who don't but have similar attributes, such as age and gender. Overall, to study a disease, it is essential to have a "disease-specific iPS cell panel" comprising patient-specific iPS cells and the patients' medical information, as well as a "healthy volunteers' iPS cell panel," comprising iPS cells derived from the cells of volunteers who do not have those diseases, and their health data. It is particularly important to obtain their health data over the long term to follow their health conditions in the future.
CiRA is establishing the patient-specific iPS cell panel using specimens and clinical information from patients suffering from specified diseases. The Hitachi Health Care Center (Hitachi City, Ibaraki Prefecture; Director: Takeshi Hayashi), which is operated by Hitachi, Ltd., will cooperate to set up the "Hitachi Healthy Volunteers' iPS Cell Panel" at CiRA by recruiting healthy volunteers visiting it.
Specifically, blood samples will be taken from consenting donors at the Hitachi Health Care Center, which will provide these samples to CiRA along with health checkup data (for which the donor's identity will remain anonymous). CiRA will then reprogram the blood cells into iPS cells to build the "Hitachi iPS cell panel."
For a healthy volunteers' iPS cell panel, it is essential to secure many healthy donorsand their health data. The Hitachi Health Care Center has experiences that its long-term health data have been utilized for the development of medical treatments. The large data at the center will enable the creation of a healthy volunteers' iPS cell panel and comparative research with the disease-specific iPS cell panel. The result is expected to contribute to understanding of the detailed causes of illnesses and diseases and the development of new therapies and pharmaceuticals.
We believe the Hitachi Health Volunteers' iPS Cell Panel will have social significance because it will become an important platform for medical applications of iPS cells. CiRA will bear the cost of organizing the Hitachi iPS cell panel.
The plan of the Hitachi iPS cell panel was approved by the Ethics Committee of Kyoto University Graduate School and Faculty of Medicine on July 27, 2015, and by the Hitachi, Ltd. Corporate Hospital Group Ethics Committee on August 17. The donor recruitment will begin in September, with the goal of collecting approximately 100 volunteers of both genders and from various ages. In cases where donors have given their consent, the iPS cells generated through the Hitachi iPS cell panel will be deposited at the Riken BioResource Center (BRC), a public cell bank, for distribution to scientists conducting iPS cell research and related applications.
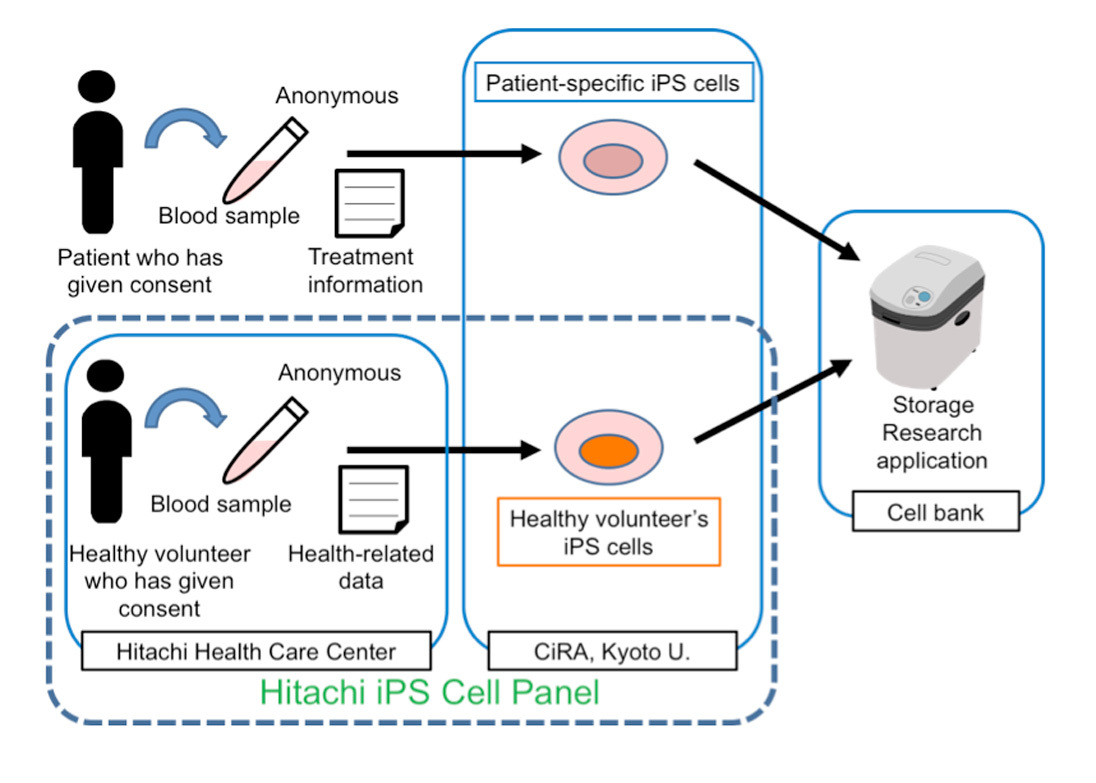
Scope of the Hitachi iPS Cell Panel
* iPS cells: Induced pluripotent stem cells. "Pluripotent" stem cells have the potential to change into any other cell type in the body (e.g., lung cells, muscle cells, or blood cells), and as such are indispensable in the field of regenerative medicine. iPS cells are pluripotent stem cells that are artificially derived from somatic cells. Dr. Shinya Yamanaka, Director of CiRA, announced the world's first successful generation of human iPS cells in 2007. Dr. Yamanaka received the Nobel Prize for Physiology or Medicine in 2012.
About Hitachi, Ltd.'s Healthcare business
Hitachi's healthcare related business involves the manufacture and sales of devices for the biomedical product (e.g., antibody pharmaceuticals, vaccines) manufacturing industry, facilities and equipment used in regenerative medicine, and devices used by hospitals and other healthcare facilities in diagnostics, testing, and treatment. In the field of regenerative medicine, the Hitachi Group provides a wide range of facilities, devices, information systems, and services, and also undertakes the development of automatic cell culturing technologies. In addition, Hitachi contributes to the development of the regenerative medicine industry through extensive collaborations with industry organizations (including the Forum for Innovative Regenerative Medicine (FIRM)) and affiliate companies. Hitachi will continue to participate in a wide range of discussions in the future, including further collaborations with CiRA in the development of cell manufacturing technologies and the use of data analysis technologies. Please refer to the Hitachi, Ltd. website for details.
About the Hitachi Health Care Center
Hitachi operates five main healthcare facilities: the Hitachi general Hospital, the Hitachinaka general Hospital, the Taga general Hospital, the Tsuchiura Medical & Health Care Center, and the Hitachi Health Care Center. The Hitachi Health Care Center in particular conducts regular health checks (including complete physical examinations) for nearly 40,000 employees of the Hitachi Group, most of whom are located in Hitachi City, Ibaraki Prefecture, as well as family members and retirees. The Center has been converting data from regular health checks into electronic format and accumulating this data for about 20 years, and is unparalleled in Japan in terms of both data volume and accuracy. Hitachi has contributed to the betterment of healthcare by effectively using data from regular health checks (with the full consent of the parties undergoing those checks) in research that has been approved by the Hitachi, Ltd. Corporate Hospital Group Ethics Committee.






















