
News & Events
News & Events
News
November 24, 2021
First in-patient transplantation of iPS cell-derived natural killer cells to treat ovarian cancer
Main Points
- Natural killer cells1) prepared from iPS cells were transplanted into an ovarian cancer patient
- The iPS cells were provided by the CiRA Foundation
- The trial is being conducted at the NCC Hospital East
The Center for iPS Cell Research and Application (CiRA), Kyoto University, and the National Cancer Center (NCC) Hospital East are cooperating on a phase I clinical trial to investigate the safety and tolerance of a new iPS cell-based adoptive cellular therapy (ACT) for ovarian cancer. The iPS cells were used to generate chimeric antigen receptor (CAR3)) innate lymphoid/natural killer cells (ILC/NK4)) targeting glypican-3 (GPC3) protein, which, for the first time, have been transplanted into a patient. CiRA Professor Shin Kaneko is leading the project.
ACT using CAR T cells and CAR NK cells has exceptional promise against cancer. Prof. Kaneko has combined CAR technologies with iPS cell technology to increase the number of patients who can benefit from this treatment. iPS cells with homologous human leukocyte antigens (HLA)5) especially are immunologically compatible with a larger number of patients. Genetic engineering of the iPS cells makes the ACT more potent.
GPC3 is an attractive target because its expression specifically correlates with several cancer cells including ovarian cancer cells. The researchers therefore genetically engineered HLA-homozygous iPS cells to express the GPC3 receptor and then differentiated the cells into NK cells for ACT.
The clinical trial began in April, and the cells were transplanted into the first patient in September.
| January 25, 2021 |
NCC Institutional Review Board approves the plan |
|---|---|
| March 12, 2021 | The plan is submitted to the Japan Pharmaceutical and Medical Devices Agency (PMDA) |
| April 12, 2021 | The clinical trial begins |
| September, 2021 | The first transplantation to a patient is administered |
(1)Name of clinical trial
A phase 1 clinical trial of intraperitoneal administration of GPC3-CAR expressing iPS cell-derived innate lymphoid/natural killer cells for patients with GPC3 positive advanced ovarian clear cell carcinoma with peritoneal dissemination.
(2)Purpose of clinical trial
To evaluate the safety and tolerance of repeated intraperitoneal injections of anti-GPC3 expressing CAR iPS cell-derived ILC/NK cells, or iCAR-ILC-N101, in patients with inoperable ovarian cancer.
(3)Cells used in the clinical trial
The iPS cell line, QHJI01s04, provided by the CiRA Foundation through its iPS cell stock2), was genetically engineered and differentiated into iCAR-ILC-N101.
(4)Target disease
Patients with inoperable and advanced ovarian follicular carcinoma expressing GPC3.
(5)About the cell transplantation
Approximately 1x106 iCAR-ILC-N101/kg are transplanted weekly for four weeks percutaneously into the peritoneal cavity. In total, the minimum and maximum doses are 0.5x106 and 3.0x106 iCAR-ILC-N101/kg, respectively.
(6)Number of patients
6-18
(7)Implementation date
April 12, 2021 - March 3, 2024
(8)Patient recruitment
For details about patient recruitment, see the NCC Hospital East site (in Japanese).
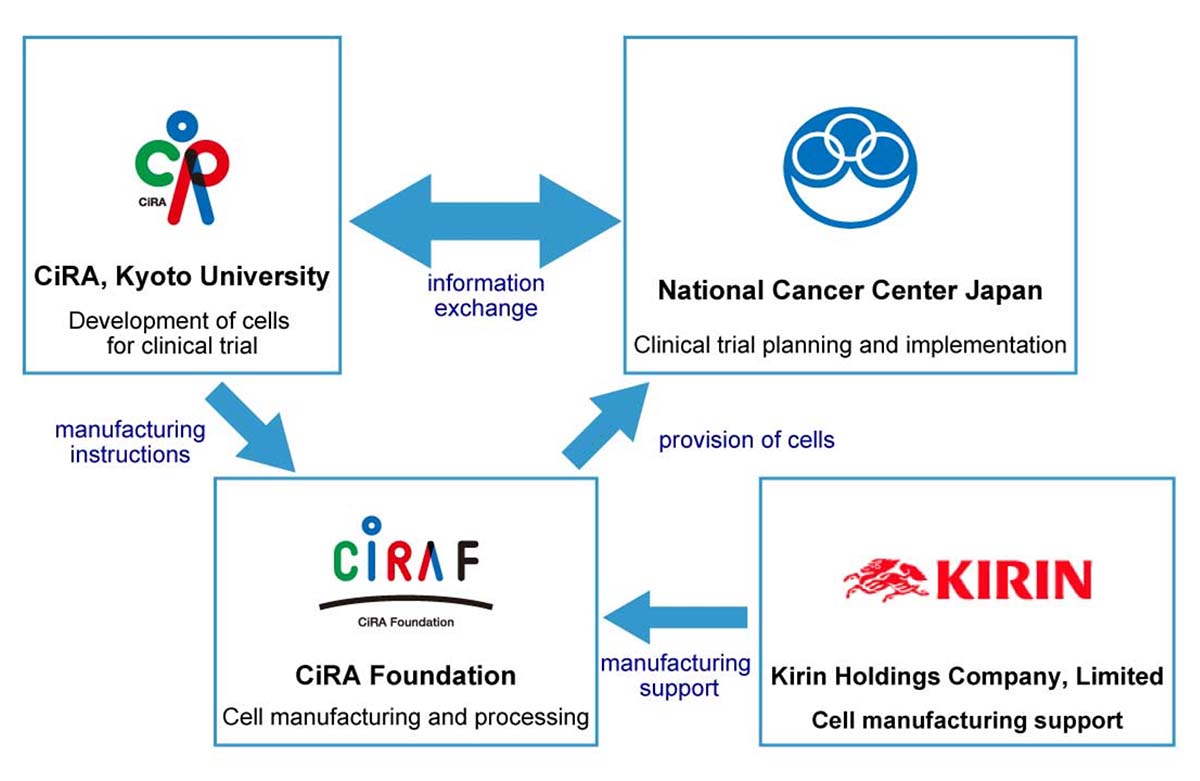
- CiRA
- Development of cells for clinical trial
- Methods for cell manufacturing and quality control
- Non-clinical safety studies
- National Cancer Center Japan
- Preparation of investigator-initiated clinical trial plan
- Implementation of trial
-
Coordinating physicians:
- Toshihiko Doi (Department of Experimental Therapeutics/Deputy Director)
- Kenichi Harano (Department of Experimental Therapeutics/Department of Medical Oncology)
- Junichiro Yuda (Department of Experimental Therapeutics/Department of Hematology)
- Akihiro Sato (Clinical Research Support Office)
National Cancer Center Hospital East-
National Cancer Center Exploratory Oncology Research & Clinical Trial Center
- Tetsuya Nakatsura (Division of Cancer Immunotherapy)
- CiRA Foundation
- Manufacturing of cellular products
- Validation of manufacturing
- Validation of analysis
- Kirin Holdings Company, Limited
- Support for manufacturing of cellular products
Organization flow chart
This research is supported by the following organizations.
- The Japan Agency for Medical Research and Development (AMED)
1) Natural killer (NK) cells
A type of cell in the innate immune system, NK cells do not respond to any specific antigens of a pathogen or cancer but rather to conditions such as cell injury.
2) iPS cell stock
The iPS cell stock at the CiRA Foundation is a collection of iPS cell lines made from HLA-homologous healthy donors available for regenerative medicine. The project began in 2013 at CiRA but was passed to the CiRA Foundation in 2020. Currently, the stock includes 27 cell lines from 7 donors and is immune compatible with 40% of the Japanese population. Through genome editing, the CiRA Foundation plans to add new iPS cell lines to increase the percentage of the compatible population.
3) Chimeric antigen receptors (CAR)
CAR are receptors that have been added to the surface of immune cells by genetic engineering. The CAR is selected based on the antigen expression of the targeted cancer cell and allows the immune cells to bind and attack this target.
4) ILC/NK
Innate lymphoid cells (ILC) are immune cells different from T cells in that they are activated independent of an antigen. However, different subsets of ILC express similar master transcription factors and cytokine profiles as different subsets of T cells. NK cells correspond to CD8 killer T cells, type 1 ILCs to type 1 CD4 helper T cells, type 2 ILCs to type 2 CD4 helper T cells, and type 3 ILCs to type 17 CD4 helper T cells. NK/ILC is a type of cell made from iPS cells that have characteristics of both ILC and NK cells.
5) HLA-homozygous iPS cells
Human antigen leukocytes (HLA) are expressed by almost all cells in the body and are used by the immune system to recognize "self" cells versus pathogenic cells. Two HLA are inherited by both parents, one from each; HLA homozygosity states that the two inherited HLA are the same. Probabilistically, HLA-homozygous cells immune-match a larger population for transplantation.






















