
News & Events
News & Events
News
April 10, 2026
Engineering the Hindbrain in a Dish to Model Development and Disease
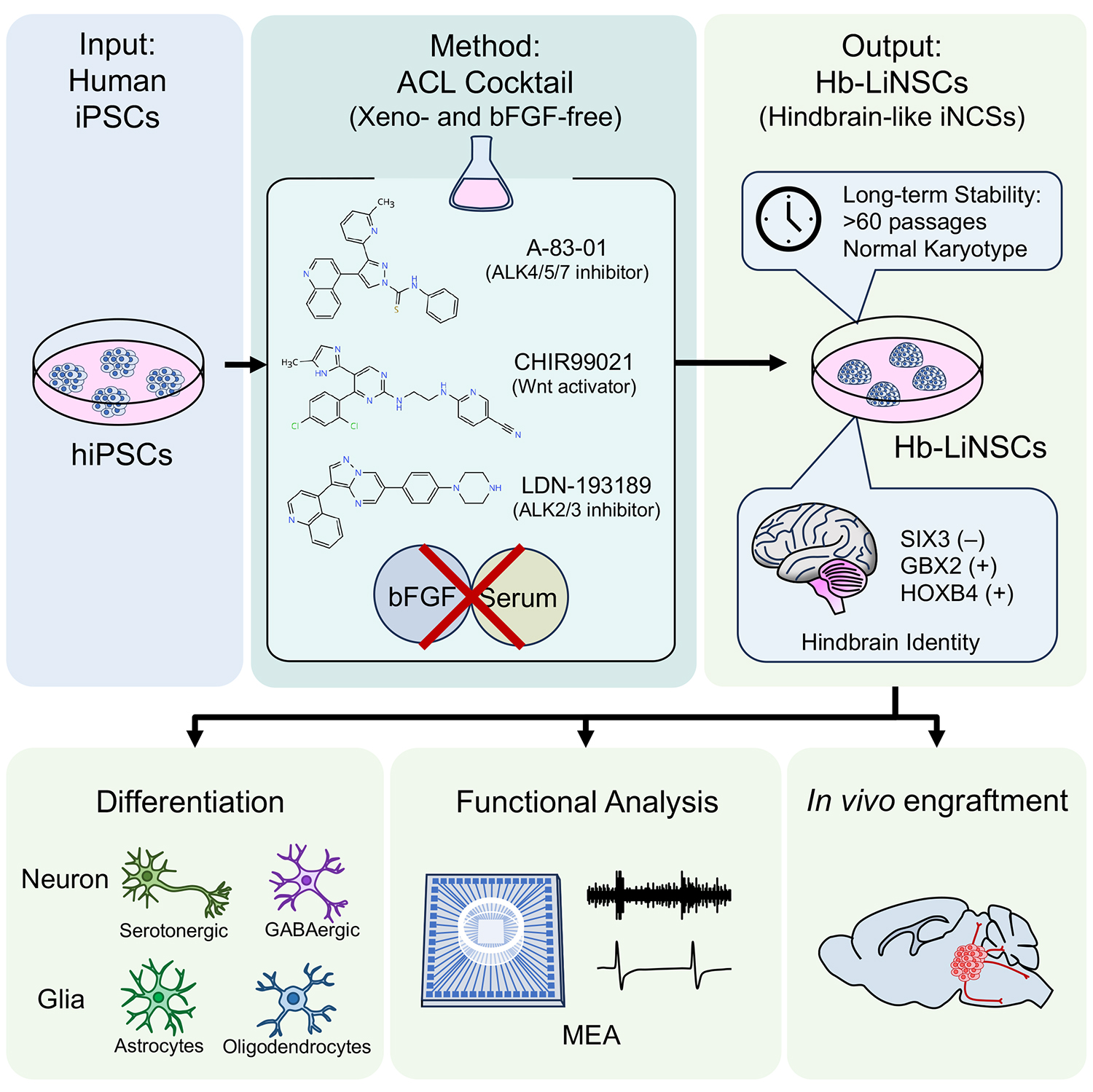
Neurons acquire distinct regional identities during development, and these identities are tightly linked to their functions and disease vulnerabilities. While neural stem cells (NSCs) derived from pluripotent stem cells are widely used in neuroscience research, preserving a stable, region-specific identity over long-term culture has remained a major challenge. To overcome these limitations, the research team devised a streamlined protocol to produce hindbrain-like induced neural stem cells (Hb LiNSCs) under minimal and defined conditions.
The newly established method relies solely on a precisely selected combination of three small molecules: High-level activation of Wnt signaling, together with dual inhibition of the SMAD pathways, directs hiPSCs away from mesodermal and endodermal fates while imparting a robust caudal, hindbrain identity. Importantly, the protocol is both xeno-free and free of basic fibroblast growth factor (bFGF), reducing variability, cost, and reliance on undefined components. Using this approach, the researchers consistently generated Hb LiNSCs from multiple hiPSC lines, demonstrating that the method is reproducible across different genetic backgrounds.
A defining feature of these Hb LiNSCs is their remarkable stability. The cells retained key neural stem cell markers, hindbrain-specific gene expression patterns, and normal chromosomal integrity for over a year of continuous culture. Transcriptomic analyses revealed that long-term maintenance did not erode neural identity or drive unwanted maturation. Instead, extended culture led to a consolidation of hindbrain characteristics, with diminishing expression of more caudal spinal cord markers over time. Single-cell RNA sequencing further confirmed that the majority of cells consistently exhibited hindbrain stem cell profiles throughout prolonged maintenance.
Beyond their stability, Hb LiNSCs remained highly functional. When differentiated in three-dimensional neurosphere cultures, the cells gave rise to multiple neural lineages, including neurons, astrocytes, and oligodendrocytes. The derived neurons formed active networks that displayed spontaneous electrical activity, as measured using multielectrode arrays, confirming functional maturation in vitro. Even after long-term expansion, these cells could be further patterned into specialized subtypes, such as serotonergic neuron progenitors, highlighting their developmental competence and flexibility.
The team also explored the behavior of these cells in vivo. Upon transplantation into mouse brains, Hb LiNSC-derived cells survived, migrated along appropriate neural pathways, and differentiated into neurons and glia, integrating into host tissue without loss of regional identity. These results underscore the physiological relevance and translational potential of the cells.
By providing an expandable, stable, and regionally defined source of human hindbrain neural stem cells, this work establishes a versatile platform for modeling hindbrain development, investigating disease mechanisms, and advancing regenerative medicine. The study represents a significant step toward more precise and scalable human neural models, bringing researchers closer to faithfully recreating the complexity of the human brain in the laboratory.
Graphical Abstract
Paper Details
- Journal: Cell Reports Methods
- Title: Wnt activation and dual SMAD inhibition for induction and maintenance of hindbrain-like neural stem cell from hiPSCs
- Authors:
Ziadoon Al-Akashi1, Denise Zujur1, Nicholas Boyd-Gibbins1, Nathalie Eileen Wiguna1, Masato Nakagawa1, Tetsuhiro Kikuchi1, Asuka Morizane1,2, Jun Takahashi1, Makoto Ikeya1,*
*: Corresponding author - Author Affiliations:
- Center for iPS Cell Research and Application (CiRA), Kyoto University
- Department of Regenerative Medicine, Center for Clinical Research and Innovation, Kobe City Medical Center General Hospital






















