
News & Events
News & Events
News
April 09, 2020
Mapping kidney development with stem cells
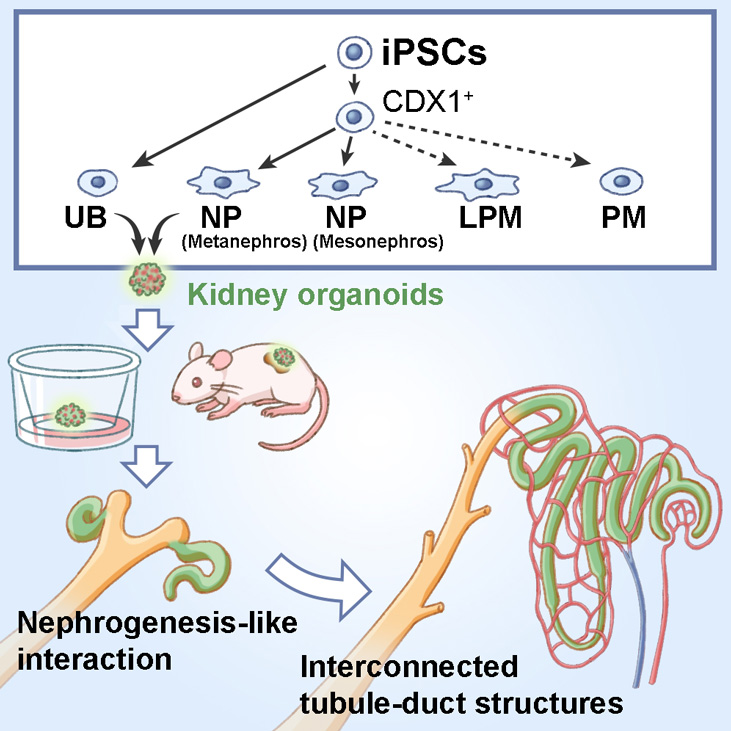
The kidney is a complicated organ that undergoes multiple stages of development. While scientists have had success recreating these stages from stem cells, doing so in just one system has proven more challenging. A new study led by CiRA Professor Kenji Osafune reports such a system with which, he says, they write a new developmental "roadmap" from human stem cells to a functional embryonic kidney. This roadmap is expected to help produce kidney cells for regenerative medicine.
Without kidney transplantation, chronic kidney disease leaves its patients with a lifetime dependency on dialysis. This has tremendous cost both in terms of economy and health, as more than 10% of the U.S. population suffers from some forms of chronic kidney disease, and the rates of dialysis are even higher in several Asian countries including Japan. Congenital kidney disease is even worse, leaving patients from birth in need of regular dialysis or renal replacement therapy. This situation has placed a priority on understanding how the kidneys develop.
Recreating the development of a functional kidney from stem cells requires the generation of several fundamental precursor cell types. Scientists have had success making most but have relied on different systems adding cost to the production.
"In the developing kidney, the mesonephros develops before the metanephros but mostly disappears and is replaced by the metanephros. We know that the kidney forms through interactions between mesonephros progenitors, metanephros progenitors, and uretic bud lineage cells, but no one can make all three of these cells using just one system," explains Osafune.
The roadmap makes clear how to acquire these three cell types from human iPS cells. The cells self-organized into kidney organoids when mixed together in a manner consistent with that seen naturally in embryonic kidney. Transplantation of the resulting kidney organoids into mice resulted in good engraftment, with blood vessels from the mice feeding the organoids.
An added discovery was that mesonephros progenitors and metanephros progenitors could be separated by adding or removing activin A, a protein with a wide range of physiological functions that go well beyond the kidney, from the differentiation protocol at a specific stage. The presence of activin A resulted in metanephros progenitors, and its absence in mesonephros progenitors.
"This was an expected finding," says Osafune. "We are still studying the reason."
By recapitulating most cell types and their interactions in kidney development, the researchers hope for their single system to benefit the study of kidney disease and therapies by simplifying experiments and lowering cost.
Paper Details
- Journal: Cell Reports
- Title: A modular differentiation system maps multiple human kidney lineages from pluripotent stem cells
- Authors: Hiraku Tsujimoto1*, Tomoko Kasahara1*, Shin-ichi Sueta1*, Toshikazu Araoka1, Satoko Sakamoto1, Chihiro Okada1,2, Shin-ichi Mae1, Taiki Nakajima1, Natsumi Okamoto1, Daisuke Taura3, Makoto Nasu1, Tatsuya Shimizu1, Makoto Ryosaka1, Zhongwei Li4, Masakatsu Sone3, Makoto Ikeya1, Akira Watanabe1, Kenji Osafune1
- Author Affiliations:
- Center for iPS Cell Research and Application (CiRA), Kyoto University, 53 Kawahara-cho, Shogoin, Sakyo-ku, Kyoto 606-8507, Japan
- Mitsubishi Space Software Co., Ltd., 5-4-36 Tsukaguchi-honmachi, Amagasaki, Hyogo 661-0001, Japan
- Department of Medicine and Clinical Science, Kyoto University Graduate School of Medicine, 54 Kawahara-cho, Shogoin, Sakyo-ku, Kyoto 606-8507, Japan
- Department of Stem Cell Biology and Regenerative Medicine, Keck School of Medicine of the University of Southern California, 1333 San Pablo Street, MMR 618, Los Angeles, CA 90033, USA






















