
Research Activities
Research Activities
Publications
July 27, 2017
Stem cells lose their shine
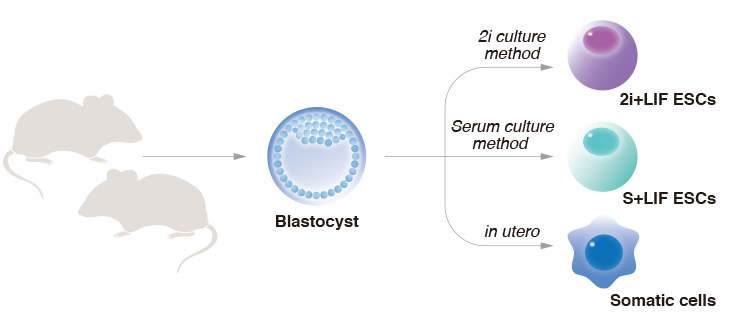
CiRA scientists show that the gold standard for preparing ES cells, the 2i culture method,
results in ES cells with abnormal epigenetics and inferior development capacity.
Because they can form any cell in the body, embryonic stem cells (ESCs) have tremendous potential for regenerative medicine. They are also expected to give great insights on the very beginnings of human development, which would explain how a small number of cells in the embryo grow into a whole body.
A new study from the CiRA laboratories of Professor Yasuhiro Yamada and Junior Associate Professor Takuya Yamamoto shows ESCs made using what Yamada calls the "gold standard method" have abnormal epigenetics that limits their usefulness in scientific research and clinical application. The findings, which can be read in Nature, suggest more attention is needed on how ESCs are made.
From a scientific perspective, a key feature of ESCs is pluripotency, which allows ESCs to grow into any type of cell. This same property is true of embryonic cells and is considered essential for development.
One way to study pluripotency is by injecting ESCs into mouse embryos. There, they mix with embryonic cells to form chimeric (genetically-modified) mice.
"When we transplant ESCs into a mouse embryo, the cells will mix with the host embryonic cells. That is how we make chimeric animals," explained Yamada. "The original ESCs were made using serum (S)+LIF. Many years later it was shown that ESCs made using 2i+LIF have better pluripotent features. These ESCs are the gold standard. The ESCs are identical genetically, but different epigenetically."
One way in which the epigenetics of 2i+LIF ESCs and S+LIF ESCs differ is in the methylation of DNA.
"DNA of 2i+LIF ESCs is more hypomethylated. Hypomethylation is an important event that happens after an egg is fertilized. The methylation levels in the embryo change with development," said Masaki Yagi, a doctoral student in the Yamada lab.
The methylation levels of 2i+LIF are thought to better recapitulate the methylation levels of embryonic cells during development, which is one reason 2i+LIF ESCs are preferred by scientists.
Like in a mouse, every gene in a mouse ESC consists of one allele from the mother and one allele from the father. For most genes, during the very earliest stages of development, demethylation will occur on both alleles. The one exception is genes within imprinting control regions. In these genes, only one allele will undergo demethylation. Having one methylated allele and one demethylated allele in the imprinting control regions is considered essential for proper development. Yagi found that in female mouse ESCs prepared in 2i+LIF, both alleles in imprinting control regions are demethylated, a phenomenon not seen in ESCs prepared in S+LIF. Moreover, this difference has a big impact on the developmental potential of the ESCs.
"We did two types of tests. One is nuclear transfer and the other is tetraploid embryo complementation. Both are stringent ways to test ESC development potential. Only S+LIF ESCs could develop into mice," said Yagi.
This finding is significant, because it forces researchers to reassess ESCs. If 2i+LIF ESCs are truly the equivalent of embryonic cells, they should have the same developmental capacity. While countless experiments have confirmed that 2i+LIF ESCs are pluripotent by creating chimeric mice, the experiments by the CiRA teams suggest the quality of the pluripotency does not match the quality in embryonic cells and that 2i+LIF ESCs may not be as well suited for the study of development as first believed.
"Because 2i+LIF ESCs successfully make chimeras, many laboratories have not considered the possibility that the pluripotency is substandard. Our findings show that scientists must select the ESC derivation protocol carefully," said Yamada.
Further experiments indicated modulating the inhibition of MEK1/2 or Src, two signaling pathways thought crucial for pluripotency, could preserve the epigenetic quality of ESCs made with 2i+LIF.
While Yagi admits most of the excitement about these findings will come from a select group of stem cell biologists, he stresses that the pluripotency quality is a major determinant in how quickly stem cell-based therapies will reach patient care.
"Our findings are helpful for generating high-quality and safer pluripotent stem cells. We demonstrated imprint-losing ESCs have lower pluripotency potential. Further research can improve stem cell derivation and propagation," Yamada said.
Paper Details
- Journal: Nature
- Title: Derivation of ground-state female ES cells maintaining gamete-derived DNA methylation
- Authors: Yagi M1, Kishigami S2, Tanaka A1, Semi K1, Mizutani E2,3, Wakayama S2,3, Wakayama T2,3, Yamamoto T1,4, and Yamada Y1
- Author Affiliations:
- Center for iPS Cell Research and Application (CiRA), Kyoto University, Kyoto, Japan
- Faculty of Life and Environmental Science, University of Yamanashi, Kofu, Japan
- Advanced Biotechnology Center, University of Yamanashi, Kofu, Japan
- AMED-CREST, AMED, Tokyo, Japan






















